The Mesoblast Ltd [ASX:MSB] posted a FY21 net loss of $98.8 million as the FDA asks for further trials of MSB’s emergency COVID treatment.
The stem cell treatments maker also admitted that it was advised to run further trials of its COVID-19 treatment before it can apply for emergency approvals.
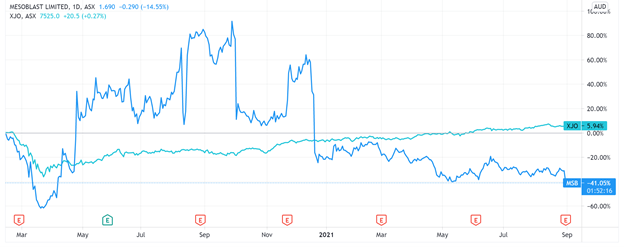
The news was poorly received by the market, with investors sending the MSB share price down 14% at noon.
Today’s drop continues a downward trend for the stock, with the MSB share price falling 65% in the last 12 months.
Mesoblast FY21 results
Total group revenue for FY21 came in at US$7.46 million, well down on the US$32.16 million recorded in FY20.
MSB said FY20 revenue included US$25 million in rexlemestrocel-L upfront and milestone payments from Grünenthal and Tasly, which was not reported in FY2021.
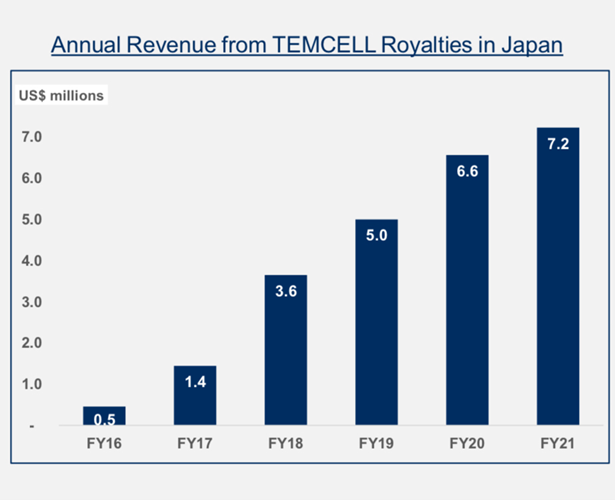
FY21 revenue from TEMCELL royalties increased by 10% from the prior year to US$7.2 million.
Mesoblast said sales of TEMCELL by its licensee for the treatment of aGVHD have ‘re-established a steady growth trajectory after plant security was expanded to meet growing demand.’
The company also cut its research and development expenses from US$56.2 million in FY21 to US$53.0 million in FY21.
The majority of MSB’s R&D spending (54%) went towards developing remestemcel-L.
The company wants its flagship remestemcel-L product approved to treat an acute immune reaction called graft-versus-host disease in children.
MSB also plans to use this same drug to treat severe respiratory distress caused by COVID-19.
The R&D cost savings were offset however, by manufacturing expenses rising $US7.4 million to US$32.7 million in FY21.
All up, the expenses contributed to the company posting a net loss after tax for FY21 of US$98.8 million, up from a net loss of US$77.9 million in the prior year.
The net loss echoed Mesoblast’s losses sustained from operating activities.
A US$30 million rise in payments to suppliers and employees saw MSB post a net cash loss from operating activities worth US$106.68 million.
This was significantly higher than the operating net cash loss of US$56.37 million the year prior.
The negative operating cash flow coupled with a net income loss were absorbed by Mesoblast raising US$106.27 million from issuing shares in FY21.
This capital injection helped MSB to end the financial year with US$136.88 million in cash and cash equivalents, up by US$6 million from FY20.
Finally, in August, MSB entered into a contractual agreement to extend the ‘interest-only period’ of its current senior debt facility to at least January 2022.
PS: We reveal four little-known small-cap stocks that cannot be ignored…Download your free report now.
Due to this, no loan payments will be needed prior to January 2022.
MSB share price outlook
A major reason for MSB’s protracted share price slump — reinforced by today’s sell-off — is the company’s mounting losses.
As Mesoblast reported in its full-year report, it has ‘incurred net losses during most of our fiscal periods since our inception.’
The FY21 net loss of $98.8 million added to an accumulated deficit worth $647.6 million.
Investors — especially ones with shorter time horizons — may have grown impatient with the stock and exited their positions.
After all, ‘When will Mesoblast turn the tide?’ they may be thinking.
It may take a while.
In the report, MSB acknowledged it ‘may continue to incur significant losses for the foreseeable future.’
These losses, predominated by research and development expenditure, could rise given the FDA requested Mesoblast to launch an additional trial of its COVID-19 treatment if it wants to apply for emergency use approvals.
Of course, the bullish counterargument is that these losses may be irrelevant in the long run once the company starts to get a return on its investment.
The medical sector is only expected to grow in line with rising global population.
MSB noted today that ‘US addressable market for acute GVHD in children and adults is ~ eight-fold larger than Japan due to greater patient numbers, incidence and pharmacoeconomics.’
Only time will tell whether this will be enough to weather Mesoblast’s foreseeable losses.
If you’re looking for more under-the-radar stocks with exciting potential but are put off by the regulatory uncertainty surrounding Mesoblast, I suggest checking out the latest briefing from our own Murray Dawes.
In a new report, Murray profiled his seven top small-cap stock ideas for 2021, including Murray’s top ASX-listed battery tech play.
If you want to read the briefing, access it here.
Regards,
Kiryll Prakapenka
For Money Morning
PS: Our publication Money Morning is a fantastic place to start on your investment journey. We talk about the big trends driving the most innovative stocks on the ASX. Learn all about it here